The IO Revolution, Part 3 of 4: The Role of the Tumor in the Cancer Immunity Cycle
Part one of this four-part series looked at how host, tumor, and environmental factors shape tumor-immune system interactions. Part two further examined the role of the host, and in this blog, we consider the role of the tumor in the cancer immunity cycle.
Tumors are highly heterogeneous, and each patient’s tumor likely represents a unique combination of tumor mutations, recruitment of immune cells, and changes to the surrounding stroma and vasculature, with concomitant alterations in metabolism, oxygenation, acidification, and nutrient availability. However, certain key traits must be acquired by all tumors as they escape intrinsic controls that normally limit unchecked cellular proliferation. These traits have been summarized as the “Hallmarks of Cancer” (Figure 1)1.
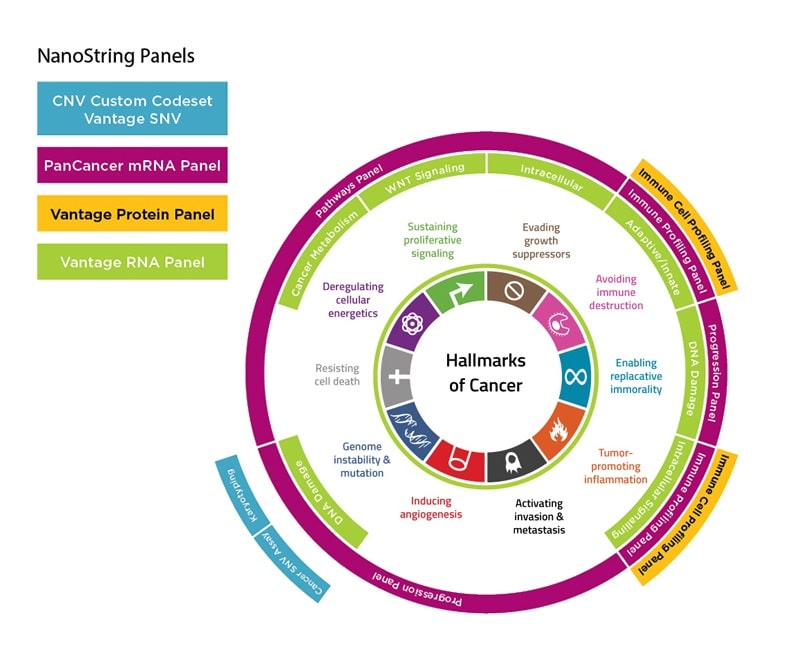
Many of these traits are tumor intrinsic activities such as altered metabolism, replicative immortality, and genomic instability. Other traits invoke points of intersection with the immune system, including promoting inflammation and avoiding immune destruction. Cancers have recently been recognized to activate signaling pathways that facilitate their evasion of immune-mediated destruction. For example, β-catenin expression in melanoma inhibits dendritic cell and T cell infiltration of the tumor by preventing the expression of CCL42. Likewise, PTEN expression in tumors induces immunosuppressive cytokine secretion, which also prevents T cell infiltration and abrogates response to T cell-mediated immunotherapy such as anti-CTLA-4 and/or anti-PD-13. Finally, epigenetic modifications within the tumor can lead to loss of Th1 trafficking cytokines, T cells, and sensitivity to checkpoint blockade4. On the other hand, mutations in DNA repair pathways can also arise spontaneously in tumors, leading to significantly increased mutational burden, and mounting evidence suggests that tumors with higher numbers of mutations are more sensitive to immunotherapies due to increased neoantigen display (Figure 2)5.

NanoString has a wide range of research tools that can provide insights into tumor biology through gene expression profiling:
- PanCancer Pathways Panel—770-plex gene expression panel addressing the 13 canonical pathways of cancer, including 124 cancer driver genes.
- PanCancer Progression Panel—770-plex gene expression panel that covers epithelial to mesenchymal transition, angiogenesis, extracellular matrix remodeling, and metastasis.
- nCounter® Vantage™ RNA Panels—192-plex gene expression panel that allows focused investigation of Wnt signaling, DNA damage and repair, and intracellular signaling, including mTOR and PI3K/AKT pathways.
- nCounter CNV Codesets—customizable CodeSets of up to 800 targets to profile replication errors that result in changes to gene copy number.
PanCancer Pathways Panel, PanCancer Progression Panel, nCounter Vantage RNA Panels, and nCounter CNV Codesets are For Research Use Only. Not for use in Diagnostic Procedures.
In the next Perspectives on Immuno-Oncology blog, we will focus on the environment-related mechanisms of immune evasion.
References
- Hanahan and Weinberg (2011) Hallmarks of cancer: the next generation. Cell 144(5):646-74.
- Spranger et al. (2015) Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity. Nature 523(7559):231-5.
- Peng et al. (2015) Loss of PTEN promotes resistance to T cell mediated immunotherapy. Cancer Discovery 6(2):202-16.
- Peng et al. (2015) Epigenetic silencing of TH1-type chemokines shapes tumour immunity and immunotherapy. Nature 527(7577):249-53.
- Alexandrov et al. (2013) Signatures of mutational processes in human cancer. Nature 500(7463):415-21.

Related Content