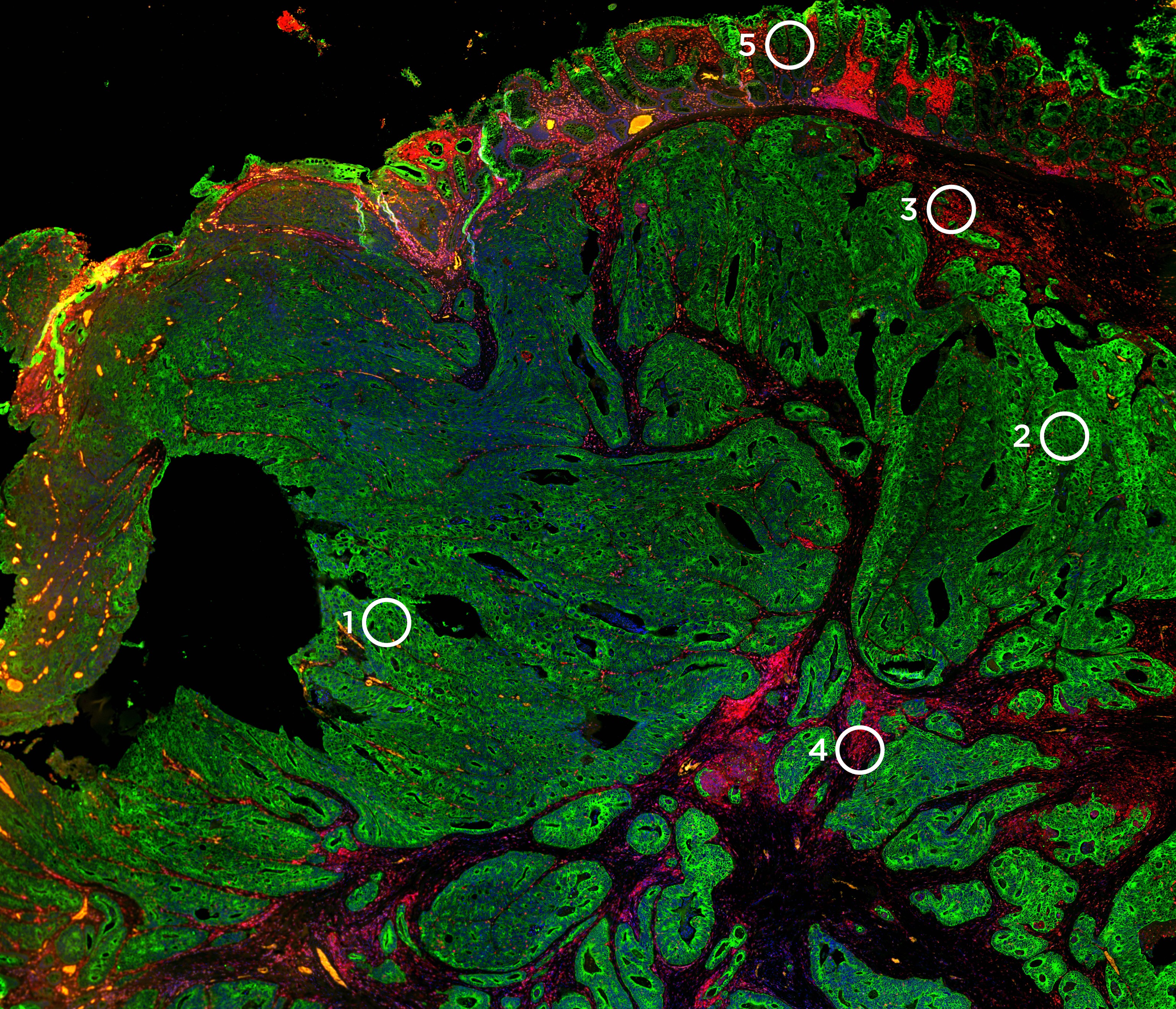
Characterizing Severe Autoimmune Toxicities Associated with Checkpoint Inhibitor Therapies
Immune checkpoint inhibitors (ICI) have emerged as powerful tools against cancer but in some cases have caused inflammation of organ systems that contributed to morbidity or mortality in patients. The molecular underpinnings of toxicities from immune checkpoint inhibitors are not well understood. To characterize the molecular features of toxicities such as colitis, myocarditis, and encephalitis, researchers at Vanderbilt University Medical Center conducted molecular analysis using digital spatial profiling. They collected healthy and afflicted tissue from patients with ICI-induced colitis and performed immunohistochemistry, RNA sequencing for over 2,000 immune-related genes, and T cell receptor sequencing to look for T cell clonality in diseased specimens.
They used digital spatial profiling technology (DSP) from NanoString to test for 20 immune-related protein biomarkers. DSP expands the technology of 3D Biology chemistry, which enables the quantitative measurement of DNA, RNA, and protein in a single sample on the same platform, so that the biomarkers can be spatially resolved. Using this technology, the researchers were able to show commonality in protein expression patterns between Crohn’s/IBD and ICI-induced colitis as well as a tendency toward greater T cell infiltrate and reduced B cell infiltrate in ICI-induced colitis specimens. Continued studies of differentially expressed cell types and gene expression markers may help better understand the disease process in ICI-induced colitis. We are currently offering a technology access program for DSP. Email tap@nanostring.com if interested in joining the program.

NanoString sat down with Justin Balko, Pharm.D., Ph.D., Assistant Professor of Medicine and Cancer Biology at Vanderbilt University Medical Center, who conducted the study, which will be presented at the upcoming American Association for Cancer Research (AACR) meeting.
NanoString (NS): Much is said about the promise of checkpoint inhibitors but not as much about severe toxicities associated with them. Is that because so little is known about them? How common are severe toxicities to immune checkpoint inhibitor (ICI) therapy?
Justin Balko (JB): Little is known about severe toxicities due to checkpoint inhibitors primarily because the agents are so new. Certainly, toxicity is much higher with the CTLA-4-targeting agents than with PD-1/L1-targeting antibodies, but toxicity can be quite prevalent when both drugs are used together. Unfortunately, the toxicities can be very sporadic, and present differently in each patient. This makes it difficult to get a perfect estimation of how frequently they are occurring in the general treated population.
NS: How did you become interested in studying this?
JB: We have been studying PD-1 therapy from a translational perspective (biomarkers of response and resistance) since I began my lab around 2014. I was working in close collaboration with Jeff Sosman and Doug Johnson, two oncologists and physician scientists at our institution (Jeff is now at Northwestern). However, in December of 2015, two cases of fatal myocarditis occurred within several weeks of one another in melanoma patients treated with the combination of anti-CTLA-4 and anti-PD-1. Given the severity of these two cases (and additional reports around the country from colleagues), we felt it was imperative to begin to understand why this was happening, so we could determine how to detect it earlier, or prevent it altogether.
NS: What did you hope to discover by doing advanced molecular analysis of healthy and afflicted tissues?
JB: We hope to identify commonalities in the features of the diseased tissues to autoimmune disorders. Making these connections will provide a good starting point to access the wealth of knowledge that has been uncovered in those areas on how to best treat the toxicities when they occur. When we first started working on these questions, we had hypotheses for what was going on in the checkpoint-inhibitor treated patients, but we needed data to support that before moving forward. Finally, we want to know what kind of immune cells are causing the damage so we can try to detect their presence earlier and design ways to reverse the inflammation.
NS: Can you explain what you found using digital spatial profiling technology?
JB: So far, we are still working through all the data (it is a lot of data!). However, we definitely see some commonalities between autoimmune diseases and the checkpoint inhibitor toxicities, but also some subtle differences.
NS: What did you find most interesting about the study’s findings?
JB: There is a striking analogy of the profiles in tissues from checkpoint inhibitor-treated patients to autoimmune disorders. However, many of the subtle differences are in things we expect to represent chronic versus acute inflammation. The checkpoint toxicities are clearly more on the acute-phase side.
NS: What are the next steps in your research?
JB: We are working on a model to study autoimmunity induced by PD-1 and CTLA-4 blockade in the lab so that we can determine the best way to therapeutically reverse it.
NS: How does this new data further our ability to predict, prevent, and treat these severe toxicities? Could this type of information potentially help stratify which patients are good candidates for ICI?
JB: The first step is for us to understand what is going on in these cases. We have a good start on that, and that will allow us to prospectively collect tissues and samples from patients going onto checkpoint therapy to see if biomarkers of autoimmune disorders are early warning signs of toxicity. We are also now developing clinical trial protocols to monitor and potentially treat these toxicities with new therapies.
If you are attending the annual AACR meeting in Washington, DC (April 1 – 5), we invite you to attend our workshop on April 3 titled, “Powering Precision Oncology Research with 3D Biology™ Technology: High Plex Multi-Analyte Profiling on FFPE with Spatial Resolution” with Gordon Mills, M.D., Ph.D., (MD Anderson Cancer Center) and Joe Beechem, Ph.D., (NanoString Technologies) presenting.
Learn more about Digital Spatial Profiling.
For Research Use Only. Not for use in diagnostic procedures.